 Use the molar mass of glucose (to one decimal place, 180. The law of conservation of mass states that matter cannot be created or destroyed, which means there must be the same number atoms at the end of a chemical reaction as at the. Step 2: Now click the button Balance to get the result. It is advisable to opt for reputable sources and ensure the calculator includes all the necessary features and functionalities. The procedure to use the balancing chemical equations calculator is as follows: Step 1: Enter the chemical equation in the input field. Stuck Review related articles/videos or use a hint. Note: All reactants and products require a coefficient of at least one. With a user-friendly interface, you can effortlessly input the unbalanced equation, and the. Balance the following chemical equation: Mg (OH) 2 + HCl MgCl 2 + H 2 O. This sophisticated tool takes the guesswork out of balancing equations. It depends on you which method you use to find this titration formula calculator. Empowering Your Calculations: The Balancing Chemical Equations Calculator: In the digital age, we have a powerful ally at our disposal - the Balancing Chemical Equations Calculator. 
The solution to this problem is to put a 2 on the left. As it is now, on the left is 1 Na, but on the right, there are 2 Na. Inspection shows that it is balanced as written, so the strategy outlined above can be adapted as follows:ġ. Use this online balance the equation calculator to convert the unbalanced equation to a balanced chemical equation that is not beneficial for productivity. Since there is an equal number of each element in the reactants and products of Na + Cl NaCl, the equation is balanced. You can directly access Balancing Chemical Equations website and get this titration calculator from here. To balance the above equation, which includes sodium and oxygen, this chemical equation must have the same amount of Na on the right side as well as on the left side. The initial step in solving a problem of this type is to write the balanced chemical equation for the reaction. If a typical 2 oz candy bar contains the equivalent of 45.3 g of glucose and the glucose is completely converted to carbon dioxide during the exam, how many grams of carbon dioxide will you produce and exhale into the exam room? Therefore, a simple, easy to use chemical equation balancer calculator is your best bet. You therefore decide to eat a candy bar to make sure that your brain does not run out of energy during the exam (even though there is no direct evidence that consumption of candy bars improves performance on chemistry exams). 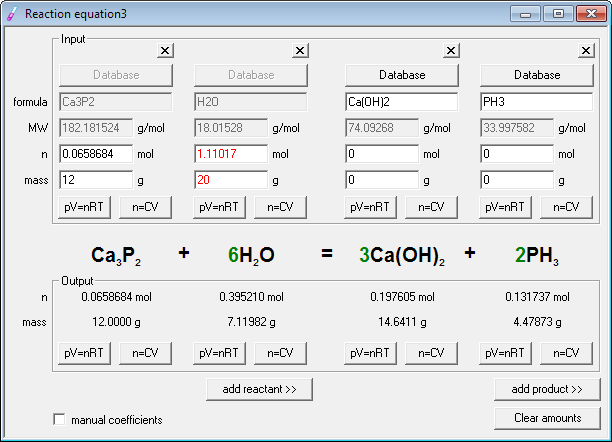
Just before a chemistry exam, suppose a friend reminds you that glucose is the major fuel used by the human brain. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
